All published articles of this journal are available on ScienceDirect.
Association between Long-COVID and Migraine: Insights From a Global Health Network Database
Abstract
Introduction
The COVID-19 pandemic has resulted in over 7 million lost lives and continues to pose a major global public health challenge. Many COVID survivors experience post-acute sequelae of SARS-CoV-2 (long-COVID). This condition presents with a wide range of symptoms such as severe fatigue, myalgia, and dyspnea. Despite growing investigation of long- COVID, its underlying pathophysiology remains unclear.
Methods
The study explored the relationship between long-COVID and migraines, including both pre-existing and newly developed, using a large global healthcare network (n = 5,187,516). Patients were categorized into long-COVID and short-COVID groups based on diagnostic criteria, and migraine prevalence was analyzed compared between groups.
Results
Patients with pre-existing migraine diagnosis had significantly higher prevalences of long-COVID (9.61%) compared to those without migraines (5.08%). Additionally, the incidence of new-onset migraine occurred more frequently among long-COVID patients (4.21%) than in the short-COVID population (1.13%).
Discussion
These findings highlight a complex association between long-COVID and migraines, suggesting potential avenues for prevention, screening, and treatment of long- COVID.
Conclusion
This study demonstrates migraine as both a predisposing factor for and a potential manifestation of long-COVID. This connection underscores the importance of recognizing migraines as part of the broader long-COVID spectrum. Improved screening for migraines and strategies aimed at autonomic regulation represent important avenues for management of long- COVID.
1. INTRODUCTION
Since the onset of the COVID-19 pandemic, more than 778 million cases have been reportedglobally [1]. Although public health efforts have decreased the overall burden of COVID-19,many patients continue to experience long-term effects of post-acute sequelae of Severe AcuteRespiratory Syndrome Coronavirus 2 (SARS-CoV-2) (long-COVID). Long-COVID can affectmultiple organ systems and commonly presents a range of symptoms, including severe fatigue,myalgias, dyspnea, and depression [2]. Long-COVID symptoms are heterogeneous and oftenfluctuate over time, making it a debilitating condition for patients and challenging for providers [3].
The pathophysiology of long-COVID remains under investigation, with proposed mechanisms including immune dysregulation, viral persistence, autonomic nervous system dysfunction, and inflammation [2,4]. One leading hypothesis is persistent sympathetic activation [5], characterized by prolonged sympathetic nervous system activation following an acute triggering event such astrauma, stress, or disease. It is emerging as a hypothesis for chronic overlapping pain conditions, including migraines, fibromyalgia, and other chronic post-infectious disorders [3,5]. Migraineand long-COVID demonstrate features of autonomic imbalance with heightened sympathetic activity and reduced parasympathetic tone [6].
Given these shared mechanisms, the study hypothesized that patients with migraines may be predisposed to developing long-COVID, and conversely, that long-COVID may increase the likelihood of new migraines, suggesting a potential bidirectional relationship. To test this, the study analyzed a global de-identified patient dataset.
2. MATERIALS AND METHODS
This retrospective cross-sectional study was conducted using TriNetX, a global health research network that aggregates de-identified electronic health records from over 124 million individuals across healthcare organizations worldwide. TriNetX was queried to identify individuals with a documented history of COVID-19 infection, with the requirement that the specified criteria be met on or after Jan 01, 2020, and the following ICD-10 codes were included: COVID-19 virus (U07.1, U07.2), COVID-19 with pneumonia (J12.82), coronavirus infection unspecified (B34.2), other coronavirus as the cause of disease classified elsewhere (B97.29), and pneumonia due to SARS-associated coronavirus (J21.81) (Fig. 1). Laboratory-confirmed SARS-CoV-2 infection was identified using curated code for positive SARS-CoV-2 and related RNA presence (+TNX Curated 9088), and SARS-CoV-2 RNA presence in respiratory system specimen (+ Logical Observation Identifiers Names and Codes (LOINC) 94500-6, +LOINC 94309-2). To isolate cases of primary SARS-CoV-2 exposure, patients with other viral infections were excluded (ICD- 9: 079.89).
From the identified COVID-19 cohort, patients were categorized into two groups: those who met criteria for long-COVID and those who did not. Long-COVID was defined with inclusion criteria of post-viral fatigue syndrome (G93.3) or malaise and fatigue (R53) recorded at least 30 days following the initial COVID-19 diagnosis and absent in the four years preceding infection (Fig. 1).
Short-COVID was defined as COVID-19 without subsequent diagnosis of fatigue or post-viral fatigue syndrome. This approach was used to minimize misclassification of chronic or unrelated fatigue symptoms. We then examined the prevalence of both pre-existing and newly diagnosed migraine (ICD-10: G43) in these groups (Fig. 1). Pre-infection migraine was defined as any migraine diagnosis recorded within one month before COVID-19 infection. New- onset migraine was defined as a diagnosis of migraine recorded within one month after COVID- 19 diagnosis in patients with no prior migraine history.
A one-month window was chosen to capture relevant migraine activity closely following SARS- CoV-2 infection, while minimizing diagnostic overlap with unrelated headache episodes. This period reflects the acute and early post-viral phase during which neuroinflammatory and autonomic changes are most likely to manifest.
Broader chronic pain conditions were not included due to their nonspecific and heterogeneous coding practices across health systems. Such terms lack standardized clinical phenotyping and may represent a wide array of conditions unrelated to migraine. Therefore, inclusion of these diagnoses could have diluted the specificity of our findings and introduced misclassification bias. Statistical analyses were performed using chi-squared analysis and verified using R statistical software version 4.5.0 (R Foundation for Statistical Computing, Vienna, Austria). Chi-squared tests with Yates’ continuity correction were used to compare the prevalence of migraine between groups. A p-value of less than 0.001 was considered statistically significant due to the large sample size.
3. RESULTS
Within the TriNetX database of over 124 million patients, a total of 5,187,516 individuals (4.11%) had a documented diagnosis of COVID-19 infection between January 1, 2020, and the time of data collection. Among the overall population, 3,304,198 individuals (2.62%) had a diagnosis of migraine, before or after their COVID-19 diagnosis.
Among patients with COVID-19 infection, 237,443 patients (4.58%) met the criteria for long- COVID, defined as the presence of new fatigue, malaise, or post-viral fatigue syndrome ≥30 days after initial infection. The remaining 4,675,106 patients made up the non–long-COVID (short-COVID) group.
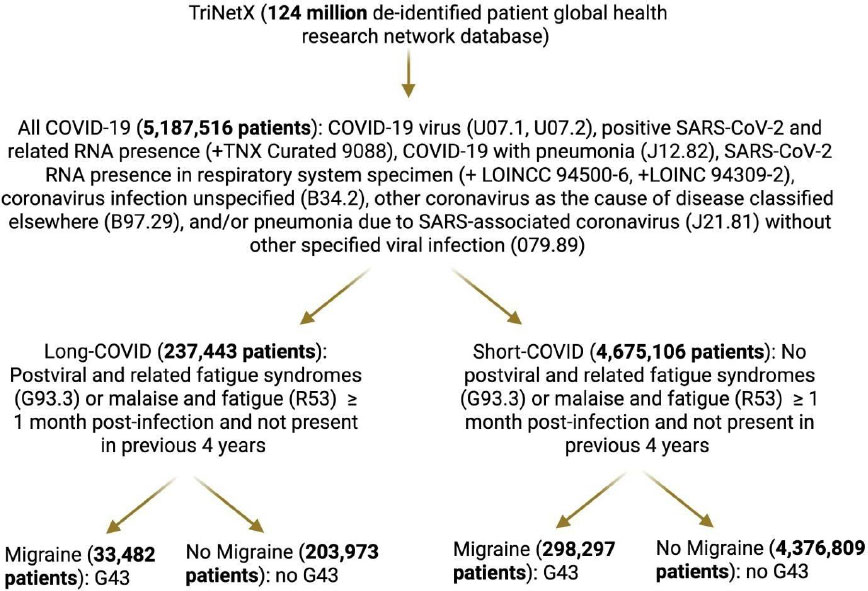
Patient selection flowchart from the TriNetX global health research network, illustrating identification of COVID-19 cases, classification into long-COVID and short-COVID cohorts, along with stratification by migraine diagnosis.
3.1. Pre-infection Migraine and Long-COVID
A greater proportion of individuals who developed long-COVID had a pre-existing diagnosis of migraine compared to those who did not develop long-COVID. Specifically, 22,813 long- COVID patients (9.61%) had migraine diagnoses prior to COVID-19 infection, in contrast to 237,487 patients (5.08%) in the non–long-COVID group.
This difference was highly statistically significant (p < 0.001), indicating a strong association between pre-existing migraine and increased risk of long-COVID development.
3.2. New-onset Migraine Post-COVID Infection
Among patients with no prior migraine diagnosis, new-onset migraine within 1 month following COVID-19 infection occurred more frequently in the long-COVID group. 300 new migraines
were observed in 9,884 patients (4.21%) in the long-COVID group compared to 53,072 patients (1.13%) in the non–long-COVID group (p < 0.001).
This nearly fourfold increase in post-infection migraine diagnoses among long-COVID patients suggests that SARS-CoV-2 infection may either trigger migraine pathophysiology or unmask a pre-existing neurologic vulnerability in susceptible individuals.
4. DISCUSSION
In this large, multi-institutional analysis, patients with long-COVID had a higher prevalence of pre-existing migraine compared to those without long-COVID (9.61% vs 5.08%). We also found that long-COVID patients were at higher risk of developing new-onset migraine compared to short-COVID patients (4.21% vs 1.13%). Together, these findings suggest a bidirectional association between migraine and long-COVID, potentially driven by shared autonomic dysregulation and sympathetic overactivation.
In this context, both migraine and long-COVID may represent different manifestations of ashared dysautonomic process, in which sympathetic hyperactivity and reduced vagal tone drive ongoing inflammation and pain sensitivity. Supporting this hypothesis, prior studies have demonstrated that post-COVID headache and migraine correlate with elevated inflammatory cytokines (Transforming Growth Factor Beta 1 and Vascular Endothelial Growth Factor) [7–9]. They have also been found to correlate with dysregulation of the renin-angiotensin system, which canincrease sympathetic tone and impair blood-brain barrier integrity [7–9]. These overlappinginflammatory and autonomic mechanisms provide a biologic explanation for the bidirectional relationship observed in our cohort.
The study's findings build on prior literature linking post-COVID headache to persistent inflammation and autonomic dysregulation. Previous meta-analyses have reported post-COVID headache in 8-15% of patients within six months of infection; however, few studies specifically examined migraine as a distinct pre-existing diagnosis [10]. Using a large, multi-institutional dataset, our study distinguishes migraine from general headache and provides quantitative evidence for a bidirectional association between migraine and long-COVID. Furthermore, the scale of the study’s analysis enables population-level support to the hypothesis that shared autonomic and inflammatory pathways underlie both disorders.
While the exact mechanisms underlying headache in long-COVID remain unclear, emerging evidence suggests that activation of trigeminal neurons and dysregulation of the angiotensin system may play central roles [10]. The Renin-Angiotensin System (RAS) can be dysregulated by SARS-CoV-2 binding to angiotensin-converting enzyme 2, leading to increased sympathetic nervous system tone and the release of inflammatory markers [9]. This dysregulation potentially contributes to the complex spectrum of long-COVID symptoms [9]. Furthermore, residual confounding variables cannot be fully excluded. Factors such as psychiatric comorbidities (e.g., anxiety and depression) and medication use may influence both migraine diagnosis and the likelihood of long-COVID. These unmeasured variables could partly account for the observed associations and should be explored in future studies.
Long-COVID patients with severe headaches have higher levels of inflammatory cytokines TGF-B1 and VEGF [ 11 ]. These inflammatory markers are also involved in microglial destruction and blood-brain barrier leakage, respectively, leading to sympathetic nervous system dysfunction [ 12 ]. Together, these findings suggest a complex interaction between inflammatory signaling in long-COVID and migraine, with chronic sympathetic activation acting as a key mediator [12,13]. In this context, SARS-CoV-2 infection may trigger migraines through direct neuroinflammatory mechanisms and may also exacerbate autonomic dysfunction in individuals with pre-existing chronic overlapping pain conditions (COPCs).
Biological sex and age may modify this relationship. Migraines are more prevalent among women, with a female-to-male ratio of 2-3:1 following puberty [14]. Sex-based differences in immune response and autonomic balance may also contribute to the higher burden of long- COVID in women [14]. Age further modifies these risks as migraine prevalence decreases after menopause and long-COVID is elevated in older adults but declines after age 70 [15]. Further studies stratified by sex and age will be important to better clarify these interactions.
The pathophysiology of long-COVID is incompletely understood. Current theories suggest mechanisms such as immune system dysregulation, virus-induced cellular changes, autonomic nervous system dysfunction, and inflammation [ 2 ]. One emerging theory proposes persistent sympathetic activation following an acute stressor such as trauma or disease [ 4 ]. This mechanism has increasingly been explored in COPCs, including migraines and other chronic post-infectious disorders [ 3 ]. Increased sympathetic activation, together with reduced parasympathetic regulation, has been described in both migraines and long-COVID [ 16 ].
Viewing COVID-19 as an immunologic insult, physiologic and psychological stressors mayexplain why COPCs, such as migraine, worsen after infection. Reduced vagal tone promotes inflammation, while vagal stimulation has been associated with symptom improvement in conditions such as rheumatoid arthritis and Crohn’s Disease [17,18]. Targeting autonomic tone, therefore, represents a promising therapeutic strategy for long-COVID [19].
4.1 Study Limitations and Strengths
This study has several limitations. One limitation is the reliance on ICD coding, which may underestimate true disease prevalence due to misclassification or coding omissions.
Specifically, the prevalence of migraine in the study (2.66% of all TriNetX patients) was lower than what is typically reported in the literature (11-15%) [20]. The definition of long-COVID, primarily based on fatigue, may have excluded other phenotypes. However, the use of standardized coding and consistent criteria across the dataset helps ensure that any misclassification would affect all groups similarly and is therefore unlikely to explain the observed differences.
The study also carries strengths. A large database that allowed analysis of 124 million patients across demographic and geographic backgrounds was used. The use of standardized definitions of migraine, long-COVID, and COVID enhances the reliability and comparability of our findings. Most importantly, our cohort definitions were conservative, using fairly restrictive terms to define each diagnosis. Broader definitions would likely have increased the associations between migraine and long-COVID. The findings may therefore underestimate the true relationship between these two conditions.
CONCLUSION
Overall, our findings suggest that migraines and long-COVID share overlapping autonomic and inflammatory pathways, supporting a bidirectional association between these conditions. Patients with long-COVID exhibited both a higher prevalence of pre-existing migraine and an increased risk of developing new-onset migraine. The study proposes that this is due to the physiologic stress of SARS-CoV-2 infection, which unmasking or exacerbates underlying autonomic imbalance. Addressing autonomic dysregulation may therefore improve long-COVID prevention, screening, and treatments.
Future studies should further investigate the contribution of other chronic overlapping pain conditions, such as fibromyalgia, chronic fatigue syndrome, and irritable bowel syndrome, as well as Postural Orthostatic Tachycardia Syndrome (POTS) and the simultaneous presence of multiple functional disorders as predisposing risk factors in the development of long-COVID.
Additionally, a deeper investigation into the demographics of patients with long-COVID and comorbid migraines may identify further contributors to these overlapping conditions. Young women have higher rates of migraine, fibromyalgia, chronic fatigue syndrome, and long-COVID. Furthermore, a retrospective cohort study discovered that Black and Hispanic patients have greater rates of COVID-19-related headaches in the United States [10]. It is vital for future research to focus on whether such demographic differences are also reflected in this global health research network. Understanding how genetic, environmental, and socioeconomic factors influence long-COVID may improve risk stratification. It is essential to highlight disparities among underserved populations and guide more equitable care strategies.
AUTHORS’ CONTRIBUTIONS
The authors confirm their contributions to the paper as follows: N.L.: Responsible for data collection, writing the paper, conceptualization, methodology, data curation, and writing the original draft; T.C. and P.K.: Contributed to the study conception and design; P.S. and L.K.: Performed data analysis and interpretation; G.C.: Contributed to conceptualization. All authors reviewed the results and approved the final version of the manuscript.
LIST OF ABBREVIATIONS
| ACE2 | = Angiotensin-Converting Enzyme 2 |
| COPC | = Chronic Overlapping Pain Condition |
| COVID-19 | = Coronavirus Disease 2019 |
| ICD | = International Classification of Diseases |
| LOINC | = Logical Observation Identifiers Names and Codes |
| POTS | = Postural Orthostatic Tachycardia Syndrome |
| RAS | = Renin-Angiotensin System |
| SARS-CoV-2 | = Severe Acute Respiratory Syndrome Coronavirus 2 |
| TGF-B1 | = Transforming Growth Factor Beta 1 |
| VEGF | = Vascular Endothelial Growth Factor |
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
The IRB determined that the proposed activity is not research involving human subjects as defined by DHHS and FDA regulations. IRB review and approval by this organization is not required.
AVAILABILITY OF DATA AND MATERIAL
All data generated or analyzed during this study are included in this published article.
ACKNOWLEDGEMENTS
Declared none.
